Rare Disease Drugs Market Size and Forecast (2025 - 2033), Global and Regional Growth, Trend, Share and Industry Analysis Report Coverage: By Therapy Type (Oncology, Hematology, Neurology, Endocrinology, Cardiovascular, Respiratory, Immunotherapy, Infectious Diseases, Others); By Drug Type (Biologics, Non-biologics); By Distribution Channel (Hospital Pharmacy, Retail Pharmacy, Online Pharmacy); and Geography
2025-08-21
Healthcare
Swetal (Research Analyst)
Description
Rare Disease Drugs Market Overview
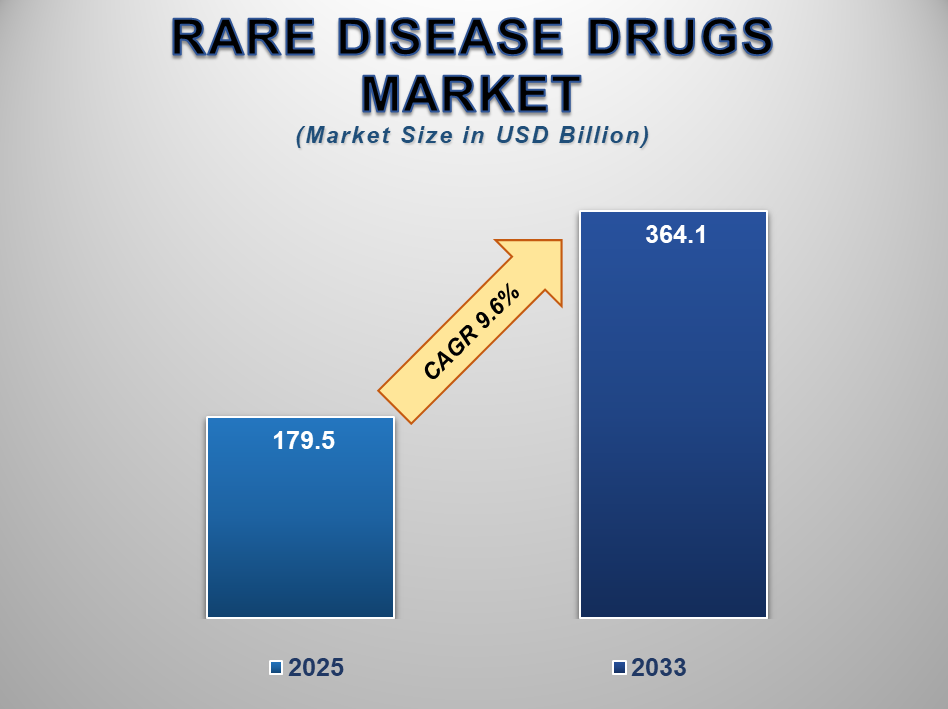
The global Rare Disease Drugs Market size is expected to grow from USD 179.5 billion in 2025 to USD 364.1 billion by 2033, expanding at a CAGR of 9.6% during the forecast period. Rare diseases, defined by their low prevalence, often lack sufficient treatment options and are frequently overlooked in traditional drug development pipelines. However, growing awareness, patient advocacy, and orphan drug incentives have driven a robust shift in R&D focus toward rare disease therapeutics.
Biopharmaceutical companies are
leveraging breakthroughs in genomics, biologics, and gene therapies to target
previously untreatable or neglected disorders. Regulatory frameworks such as
the U.S. Orphan Drug Act and similar incentives in the EU and Japan have
accelerated drug approvals, offering extended market exclusivity and financial
benefits. Additionally, rising diagnostic rates and personalized medicine
approaches have fueled earlier and more accurate detection of rare conditions,
increasing treatment accessibility. With supportive policy structures and
innovation in therapeutic platforms, the rare disease drug landscape is
entering a phase of transformative growth.
Rare Disease Drugs Market
Drivers and Opportunities
Rising R&D investments and orphan drug designations are
anticipated to lift the Rare Disease Drugs market during the forecast period
The rising prevalence of rare
diseases, now estimated to affect over 300 million people globally, has
prompted an urgent focus on drug discovery and development. Governments and
regulatory agencies worldwide have implemented orphan drug frameworks that
incentivize pharmaceutical companies through grants, tax credits, accelerated
approval pathways, and market exclusivity extensions. These favorable policies
have helped shift drug developers’ attention toward small-patient-population
diseases that were previously commercially unviable. Large pharmaceutical firms
and biotech startups alike are increasing their investment in rare disease
pipelines, targeting neurological, hematological, and oncological conditions
with high unmet needs. Innovative approaches like CRISPR-based gene editing,
antisense oligonucleotides (ASOs), RNA-based therapeutics, and monoclonal
antibodies are significantly enhancing the effectiveness and precision of rare
disease treatments. Furthermore, collaborations between industry, academia, and
patient advocacy groups are reducing R&D bottlenecks and improving trial
outcomes. The convergence of regulatory support and scientific advancement is
expected to continue driving rare disease drug innovation and market growth.
Rapid adoption of precision medicine and advanced diagnostics
is anticipated to lift the Rare Disease Drugs market during the forecast period
Technological progress in
genomics, bioinformatics, and diagnostic tools is transforming how rare
diseases are identified and treated. Next-generation sequencing (NGS),
whole-exome sequencing, and molecular diagnostics are enabling earlier and more
accurate diagnoses, shortening the typical diagnostic odyssey that rare disease
patients often face. With quicker identification of specific genetic mutations,
treatment decisions can be tailored to the individual, optimizing outcomes and
reducing the burden of trial-and-error prescribing. This shift toward precision
medicine is especially impactful for rare diseases that present heterogeneously
or mimic more common conditions. As genetic screening becomes more affordable
and accessible, even healthcare systems in emerging economies are beginning to
integrate these tools. Pharma and biotech companies are concurrently developing
targeted therapies, particularly biologics and gene therapies, tailored to
specific genetic profiles. This personalized approach not only improves
efficacy but also meets increasing patient expectations for customized care.
The synergy between precision diagnostics and individualized therapies is
expected to be a major growth accelerator for the rare disease drug market over
the next decade.
Opportunity for the Rare Disease Drugs Market
Rising demand for targeted biologics and gene therapies
creates a significant opportunity in the global Rare Disease Drugs market
The global shift toward biologic
drugs and gene therapies is unlocking new therapeutic avenues for rare diseases
that were previously untreatable. Biologics, including monoclonal antibodies
and enzyme replacement therapies, offer high specificity and efficacy in
managing complex conditions such as lysosomal storage disorders, spinal
muscular atrophy, and certain rare cancers. Simultaneously, the approval of
breakthrough gene therapies has provided long-term, potentially curative
options for diseases like Duchenne muscular dystrophy and thalassemia. This
rising demand for high-value, targeted therapies is attracting increased
investment and partnership activity across the life sciences sector. Venture
capital firms, pharmaceutical giants, and specialized biotech companies are
heavily funding R&D pipelines focused on rare disease biologics and gene
delivery systems. Moreover, favorable reimbursement policies and global efforts
to streamline regulatory pathways are supporting faster market entry. Rising demand
for targeted biologics and gene therapies creates a significant opportunity in
the global Rare Disease Drugs market, reshaping therapeutic strategies and
long-term disease management worldwide.
Rare Disease Drugs Market Scope
|
Report Attributes |
Description |
|
Market Size in 2025 |
USD 179.5 Billion |
|
Market Forecast in 2033 |
USD 364.1 Billion |
|
CAGR % 2025-2033 |
9.6% |
|
Base Year |
2024 |
|
Historic Data |
2020-2024 |
|
Forecast Period |
2025-2033 |
|
Report USP |
Production, Consumption, company share, company heatmap, company
production Capacity, growth factors and more |
|
Segments Covered |
●
By Therapy Type ●
By Drug Type ●
By Distribution Channel |
|
Regional Scope |
●
North America ●
Europe ●
APAC ●
Latin America ●
Middle East and Africa |
|
Country Scope |
1)
U.S. 2)
Canada 3)
Germany 4)
UK 5)
France 6)
Spain 7)
Italy 8)
Switzerland 9)
China 10)
Japan 11)
India 12)
Australia 13)
South Korea 14)
Brazil 15)
Mexico 16)
Argentina 17)
South Africa 18)
Saudi Arabia 19) UAE |
The global Rare Disease Drugs
Market is segmented by Therapy Type, by Drug Type, by Distribution Channel, and
by Region.
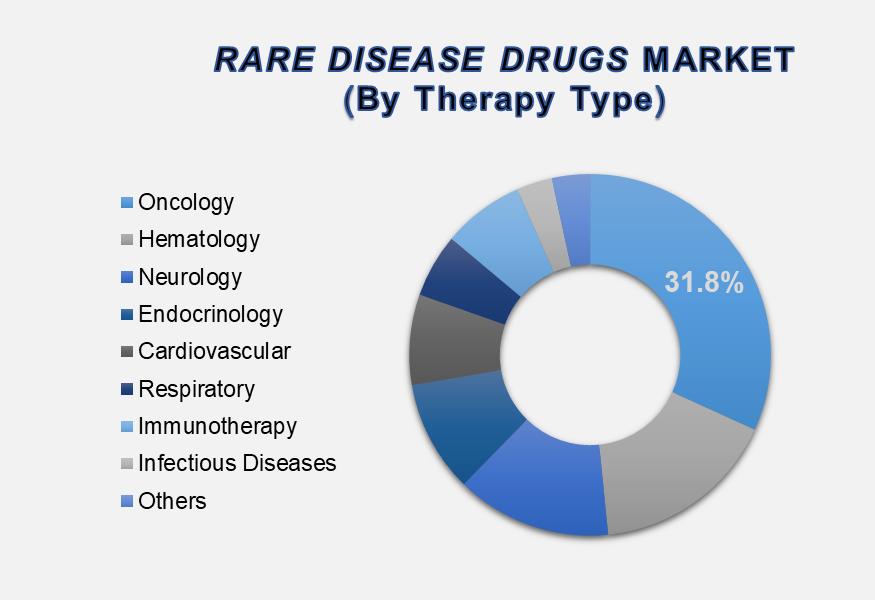
The Oncology segment accounted for the largest market share
in the global Rare Disease Drugs market
By Therapy Type, the market includes Oncology, Hematology, Neurology, Endocrinology, Cardiovascular, Respiratory, Immunotherapy, Infectious Diseases, and Others. Among these, the Oncology segment held the dominant position in 2024, contributing 31.8% of the global market share. This dominance is driven by the high incidence of rare forms of cancer, such as multiple myeloma, sarcomas, and certain pediatric malignancies that lack effective treatment options. The introduction of orphan oncology drugs and targeted biologics, along with strong support from regulatory agencies for expedited approvals, has accelerated development in this category. Additionally, pharmaceutical companies are actively investing in immuno-oncology and gene-based treatments for rare cancer subtypes. With continued innovation in precision oncology and increased awareness about rare cancers, the segment is projected to sustain its leading position throughout the forecast period.
The Biologics segment accounted for the largest market share
in the global Rare Disease Drugs market
By Drug Type, the market is
bifurcated into Biologics and Non-biologics. The Biologics segment leads due to
its proven efficacy in treating complex, life-threatening rare conditions such
as spinal muscular atrophy, lysosomal storage disorders, and rare cancers.
Biologics include monoclonal antibodies, enzyme replacement therapies, and cell
and gene therapies that offer targeted mechanisms of action with fewer
off-target effects. The segment is also bolstered by increasing FDA and EMA
approvals of biologic orphan drugs and expanding investments from biotech and
large pharmaceutical companies. Moreover, the advancements in biomanufacturing
and the ability to tailor therapies to specific genetic markers support the
broader adoption of biologics. As more gene and RNA-based therapies enter
late-stage pipelines, the biologics segment is expected to continue dominating
the rare disease drugs landscape.
The Hospital Pharmacy segment accounted for the largest
market share in the global Rare Disease Drugs market
By Distribution Channel, the
market is categorized into Hospital Pharmacy, Retail Pharmacy, Online Pharmacy,
and Others. The Hospital Pharmacy segment held the largest share in 2024 due to
the complex nature and high cost of rare disease drugs, which often require
specialized handling and administration under medical supervision. Many rare
disease treatments, particularly biologics and injectables, are administered
intravenously or require cold chain logistics, making hospital settings the
preferred point of distribution. In addition, hospitals provide better
infrastructure for diagnostics, monitoring, and patient education related to
these therapies. With the increasing use of personalized medicine and
hospital-based gene therapy centers, hospital pharmacies are playing a central
role in rare disease treatment delivery. This trend is anticipated to continue,
maintaining the dominance of this segment through 2033.
The following segments are part
of an in-depth analysis of the global Rare Disease Drugs Market:
|
Market Segments |
|
|
By Therapy Type |
●
Oncology ●
Hematology ●
Neurology ●
Endocrinology ●
Cardiovascular ●
Respiratory ●
Immunotherapy ●
Infectious Diseases ●
Others |
|
By Drug Type |
●
Biologics ●
Non-biologics |
|
By Distribution Channel |
●
Hospital Pharmacy ●
Retail Pharmacy ●
Online Pharmacy ●
Others |
Rare Disease Drugs Market
Share Analysis by Region
The North America region is projected to hold the largest
share of the global Rare Disease Drugs market over the forecast period.
North America accounted for the
largest share of the global Rare Disease Drugs market in 2024, supported by
favorable regulatory frameworks such as the Orphan Drug Act, strong research
infrastructure, and high healthcare expenditure. The U.S., in particular, has
been at the forefront of rare disease drug development, with a high number of
orphan drug designations and approvals by the FDA. The presence of leading
pharmaceutical companies, early adoption of gene and cell therapies, and robust
insurance coverage for high-cost drugs have further contributed to the region’s
dominance. Furthermore, collaborations between government bodies, industry
stakeholders, and patient advocacy organizations have created an ecosystem
conducive to rare disease innovation and patient support.
Asia Pacific is poised to
register the highest growth rate during the forecast period, driven by
improving diagnostic capabilities, rising awareness, and government efforts to
develop national rare disease registries. Countries like China, Japan, and India
are expanding their investments in biotechnology and rare disease research.
Japan’s “Strategy for Rare and Intractable Diseases” and China’s inclusion of
rare diseases in national reimbursement lists are examples of strategic policy
moves supporting market expansion. The region also benefits from a growing
pharmaceutical manufacturing base and clinical trial activity, making it a
high-potential geography for future growth in rare disease therapeutics.
Rare Disease Drugs Market Competition Landscape Analysis
The Rare
Disease Drugs market is characterized by strong participation from global
pharmaceutical and biotech companies with deep R&D pipelines and
significant investments in biologics and gene therapies. Leading players
include Novartis, Roche, Sanofi, Pfizer, Bristol Myers Squibb, and Johnson
& Johnson, who have all launched or are developing breakthrough therapies
across oncology, hematology, and neurology segments.
Global Rare Disease Drugs
Market Recent Developments News:
- In February
2024 – Rapid Commercialization Partners (RCP) formed a strategic alliance
with Orphan Now to enhance orphan drug development and accelerate global
patient access to rare disease treatments through optimized
commercialization pathways.
- In February 2023 – GSK plc obtained FDA Orphan Drug
Designation for Benlysta (belimumab), its B-cell inhibiting monoclonal
antibody for systemic sclerosis treatment. The company announced plans to
commence a phase II/III trial for systemic sclerosis-associated
interstitial lung disease (SSc-ILD) in H1 2023.
- In December 2022 – Amgen Inc. executed a landmark
$27.8 billion acquisition of Horizon Therapeutics Plc, significantly
expanding its rare disease therapeutic portfolio through Horizon's
established orphan drug pipeline.
- In November 2022 – Alexion, AstraZeneca's rare disease unit,
strengthened its genomic medicine capabilities by acquiring LogicBio
Therapeutics, Inc., a leader in innovative genetic therapies for rare
disorders.
The Global Rare Disease
Drugs Market is dominated by a few large companies, such as
●
Novartis
●
Roche
●
Sanofi
●
Pfizer
●
Bristol Myers Squibb
(BMS)
●
Johnson & Johnson
(Janssen)
●
Takeda Pharmaceutical
●
AstraZeneca
●
Merck & Co. (MSD)
●
Amgen
●
Vertex Pharmaceuticals
●
Biogen
●
Alexion
Pharmaceuticals (AstraZeneca Rare Disease)
●
CSL Behring
●
Ultragenyx
Pharmaceutical
●
BioMarin
Pharmaceutical
●
Sarepta Therapeutics
●
Ionis Pharmaceuticals
●
Regeneron
Pharmaceuticals
●
bluebird bio
● Other Prominent Players
Frequently Asked Questions
Swetal (Research Analyst)
Swetal is a Research Analyst with 4+ years of experience specializing in healthcare market research, medical devices, healthcare IT, and medical technology industry analysis. Her expertise includes competitive intelligence, secondary research, market trend forecasting, and healthcare business insights across global healthcare ecosystems.
She contributes to advanced market research reports by analyzing regulatory frameworks, emerging healthcare technologies, competitive landscapes, and innovation trends shaping the medical industry. With a strong focus on accurate market intelligence, strategic insights, and healthcare industry trends, Swetal supports businesses in making informed, data-backed decisions in a rapidly transforming healthcare environment.
- Global Rare Disease Drugs Market Introduction and Market Overview
- Objectives of the Study
- Global Rare Disease Drugs Market Scope and Market Estimation
- Global Rare Disease Drugs Market Overall Market Size (US$ Bn), Market CAGR (%), Market forecast (2025 - 2033)
- Global Rare Disease Drugs Market Revenue Share (%) and Growth Rate (Y-o-Y) from 2021 - 2033
- Market Segmentation
- Therapy Type of Global Rare Disease Drugs Market
- Drug Type of Global Rare Disease Drugs Market
- Distribution Channel of Global Rare Disease Drugs Market
- Region of Global Rare Disease Drugs Market
- Executive Summary
- Demand Side Trends
- Key Market Trends
- Market Demand (US$ Bn) Analysis 2021 – 2024 and Forecast, 2025 – 2033
- Demand and Opportunity Assessment
- Demand Supply Scenario
- Market Dynamics
- Drivers
- Limitations
- Opportunities
- Impact Analysis of Drivers and Restraints
- Emerging Trends for Rare Disease Drugs Market
- Porter’s Five Forces Analysis
- PEST Analysis
- Key Regulation
- Global Rare Disease Drugs Market Estimates & Historical Trend Analysis (2021 - 2024)
- Global Rare Disease Drugs Market Estimates & Forecast Trend Analysis, by Therapy Type
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by Therapy Type, 2021 - 2033
- Oncology
- Hematology
- Neurology
- Endocrinology
- Cardiovascular
- Respiratory
- Immunotherapy
- Infectious Diseases
- Others
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by Therapy Type, 2021 - 2033
- Global Rare Disease Drugs Market Estimates & Forecast Trend Analysis, by Drug Type
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by Drug Type, 2021 - 2033
- Biologics
- Non-biologics
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by Drug Type, 2021 - 2033
- Global Rare Disease Drugs Market Estimates & Forecast Trend Analysis, by Distribution Channel
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by Distribution Channel, 2021 - 2033
- Hospital Pharmacy
- Retail Pharmacy
- Online Pharmacy
- Others
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by Distribution Channel, 2021 - 2033
- Global Rare Disease Drugs Market Estimates & Forecast Trend Analysis, by region
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by region, 2021 - 2033
- North America
- Europe
- Asia Pacific
- Middle East & Africa
- Latin America
- Global Rare Disease Drugs Market Revenue (US$ Bn) Estimates and Forecasts, by region, 2021 - 2033
- North America Rare Disease Drugs Market: Estimates & Forecast Trend Analysis
- North America Rare Disease Drugs Market Assessments & Key Findings
- North America Rare Disease Drugs Market Introduction
- North America Rare Disease Drugs Market Size Estimates and Forecast (US$ Billion) (2021 - 2033)
- By Therapy Type
- By Drug Type
- By Distribution Channel
- By Country
- The U.S.
- Canada
- North America Rare Disease Drugs Market Assessments & Key Findings
- Europe Rare Disease Drugs Market: Estimates & Forecast Trend Analysis
- Europe Rare Disease Drugs Market Assessments & Key Findings
- Europe Rare Disease Drugs Market Introduction
- Europe Rare Disease Drugs Market Size Estimates and Forecast (US$ Billion) (2021 - 2033)
- By Therapy Type
- By Drug Type
- By Distribution Channel
- By Country
- Germany
- Italy
- K.
- France
- Spain
- Switzerland
- Rest of Europe
- Europe Rare Disease Drugs Market Assessments & Key Findings
- Asia Pacific Rare Disease Drugs Market: Estimates & Forecast Trend Analysis
- Asia Pacific Market Assessments & Key Findings
- Asia Pacific Rare Disease Drugs Market Introduction
- Asia Pacific Rare Disease Drugs Market Size Estimates and Forecast (US$ Billion) (2021 - 2033)
- By Therapy Type
- By Drug Type
- By Distribution Channel
- By Country
- China
- Japan
- India
- Australia
- South Korea
- Rest of Asia Pacific
- Asia Pacific Market Assessments & Key Findings
- Middle East & Africa Rare Disease Drugs Market: Estimates & Forecast Trend Analysis
- Middle East & Africa Market Assessments & Key Findings
- Middle East & Africa Rare Disease Drugs Market Introduction
- Middle East & Africa Rare Disease Drugs Market Size Estimates and Forecast (US$ Billion) (2021 - 2033)
- By Therapy Type
- By Drug Type
- By Distribution Channel
- By Country
- South Africa
- UAE
- Saudi Arabia
- Rest of MEA
- Middle East & Africa Market Assessments & Key Findings
- Latin America Rare Disease Drugs Market: Estimates & Forecast Trend Analysis
- Latin America Market Assessments & Key Findings
- Latin America Rare Disease Drugs Market Introduction
- Latin America Rare Disease Drugs Market Size Estimates and Forecast (US$ Billion) (2021 - 2033)
- By Therapy Type
- By Drug Type
- By Distribution Channel
- By Country
- Brazil
- Mexico
- Argentina
- Rest of LATAM
- Latin America Market Assessments & Key Findings
- Country Wise Market: Introduction
- Competition Landscape
- Global Rare Disease Drugs Market Product Mapping
- Global Rare Disease Drugs Market Concentration Analysis, by Leading Players / Innovators / Emerging Players / New Entrants
- Global Rare Disease Drugs Market Tier Structure Analysis
- Global Rare Disease Drugs Market Concentration & Company Market Shares (%) Analysis, 2023
- Company Profiles
- Novartis
- Company Overview & Key Stats
- Financial Performance & KPIs
- Product Portfolio
- SWOT Analysis
- Business Strategy & Recent Developments
- Novartis
* Similar details would be provided for all the players mentioned below
- Roche
- Sanofi
- Pfizer
- Bristol Myers Squibb (BMS)
- Johnson & Johnson (Janssen)
- Takeda Pharmaceutical
- AstraZeneca
- Merck & Co. (MSD)
- Amgen
- Vertex Pharmaceuticals
- Biogen
- Alexion Pharmaceuticals (AstraZeneca Rare Disease)
- CSL Behring
- Ultragenyx Pharmaceutical
- BioMarin Pharmaceutical
- Sarepta Therapeutics
- Ionis Pharmaceuticals
- Regeneron Pharmaceuticals
- bluebird bio
- Other Prominent Players
- Research Methodology
- External Transportations / Databases
- Internal Proprietary Database
- Primary Research
- Secondary Research
- Assumptions
- Limitations
- Report FAQs
- Research Findings & Conclusion
Our Research Methodology
"Insight without rigor is just noise."
We follow a comprehensive, multi-phase research framework designed to deliver accurate, strategic, and decision-ready intelligence. Our process integrates primary and secondary research , both quantitative and qualitative , along with dual modeling techniques ( top-down and bottom-up) and a final layer of validation through our proprietary in-house repository.
PRIMARY RESEARCH
Primary research captures real-time, firsthand insights from the market to understand behaviors, motivations, and emerging trends.
1. Quantitative Primary Research
Objective: Generate statistically significant data directly from market participants.
Approaches:- Structured surveys with customers, distributors, and field agents
- Mobile-based data collection for point-of-sale audits and usage behavior
- Phone-based interviews (CATI) for market sizing and product feedback
- Online polling around industry events and digital campaigns
- Purchase frequency by customer type
- Channel performance across geographies
- Feature demand by application or demographic
2. Qualitative Primary Research
Objective: Explore decision-making drivers, pain points, and market readiness.
Approaches:- In-depth interviews (IDIs) with executives, product managers, and key decision-makers
- Focus groups among end users and early adopters
- Site visits and observational research for consumer products
- Informal field-level discussions for regional and cultural nuances
SECONDARY RESEARCH
This phase helps establish a macro-to-micro understanding of market trends, size, regulation, and competitive dynamics, sourced from credible and public domain information.
1. Quantitative Secondary Research
Objective: Model market value and segment-level forecasts based on published data.
Sources include:- Financial reports and investor summaries
- Government trade data, customs records, and regulatory statistics
- Industry association publications and economic databases
- Channel performance and pricing data from marketplace listings
- Revenue splits, pricing trends, and CAGR estimates
- Supply-side capacity and volume tracking
- Investment analysis and funding benchmarks
2. Qualitative Secondary Research
Objective: Capture strategic direction, innovation signals, and behavioral trends.
Sources include:- Company announcements, roadmaps, and product pipelines
- Publicly available whitepapers, conference abstracts, and academic research
- Regulatory body publications and policy briefs
- Social and media sentiment scanning for early-stage shifts
- Strategic shifts in market positioning
- Unmet needs and white spaces
- Regulatory triggers and compliance impact
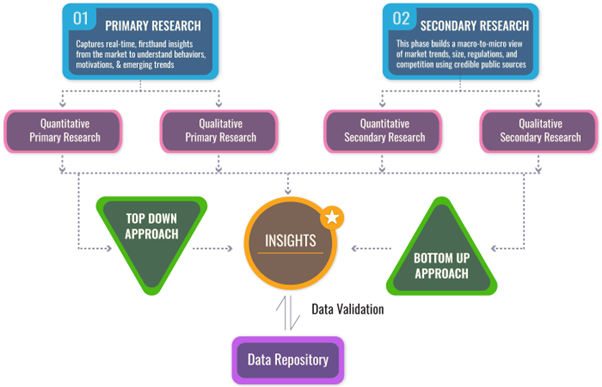
DUAL MODELING: TOP-DOWN + BOTTOM-UP
To ensure robust market estimation, we apply two complementary sizing approaches:
Top-Down Modeling:- Start with broader industry value (e.g., global or regional TAM)
- Apply filters by segment, geography, end-user, or use case
- Adjust with primary insights and validation benchmarks
- Ideal for investor-grade market scans and opportunity mapping
- Aggregate from the ground up using sales volumes, pricing, and unit economics
- Use internal modeling templates aligned with stakeholder data
- Incorporate distributor-level or region-specific inputs
- Most accurate for emerging segments and granular sub-markets
DATA VALIDATION: IN-HOUSE REPOSITORY
We close the loop with proprietary data intelligence built from ongoing projects, industry monitoring, and historical benchmarking. This repository includes:
- Multi-sector market and pricing models
- Key trendlines from past interviews and forecasts
- Benchmarked adoption rates, churn patterns, and ROI indicators
- Industry-specific deviation flags and cross-check logic
- Catches inconsistencies early
- Aligns projections across studies
- Enables consistent, high-trust deliverables

